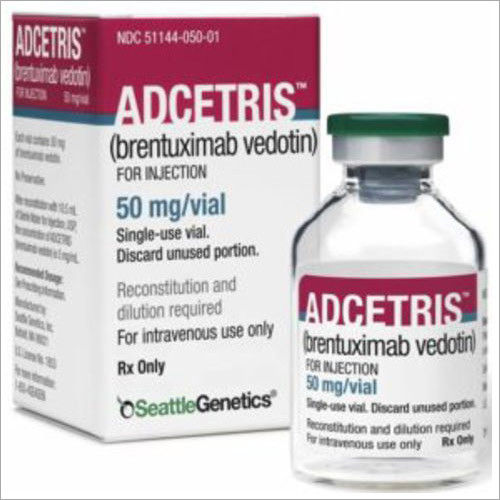
Brentuximab Vedotin Injection
Product Details:
X
Product Description
Technical Details:- Brand Name: Adcetris
- Manufacturer: Seattle Genetics
- Ingredients: Brentuximab vedotin
- Type: Injection
- Power: 50 mg/vial
Brentuximab vedotin (INN, trade name Adcetris) is an antibody-drug conjugate medication used for the treatment of relapsed or refractory Hodgkin lymphoma (HL) and systemic anaplastic large cell lymphoma (ALCL). It selectively targets tumor cells expressing the CD30 antigen, a defining marker of Hodgkin lymphoma and ALCL (a type of T cell non-Hodgkin lymphoma).
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese
 Call Me Free
Call Me Free